|
|
|
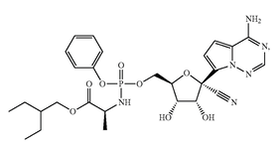
Wuhan Institute of Virology filed a patent application at the Chinese patent office on the use of Gilead’s Remdesivir for treating COVID-19 on January 21, 2020. The patent application raises questions of how could Wuhan file a patent application to something (in this case, a drug) that does not belong to them. Many people have a difficult time understanding how a third-party can patent something using a product belonging to someone else. To understand why this is the case, you must first understand that a patent is a negative right -- it is not a positive right. As such, a patent does not allow the owner to make, use, offer to sell, or sell the invention. A patent merely allows the owner to prevent others from doing the same. There may be other patents or regulatory hurdles blocking the patent owner from actually making or using his own product. In other words, just because you get a patent, does not mean that you can actually commercialize your product. There is a difference between patenting a product and having freedom to operate (FTO) to use that product. I’ve discussed the differences between patentability and FTO in previous posts, but here I will discuss it using Wuhan’s patent application to using Remdesivir for treating COVID-19 as an example. Gildead’s Patents for Remdesivir Gilead has several patents covering its antiviral drug, Remdesivir. For the purposes of this post, I will only examine two. Gilead filed U.S. patent application number 14/613,719 on February 4, 2015 which claimed priority to U.S. Provisional Patent Application Number 61/366,609 filed on July 22, 2010. This application issued on September 4, 2018 as U.S. Patent Number 10,065,958 (the ‘958 patent) with independent claim 1 broadly directed to the composition as follows: 1.A compound that is Gilead also filed U.S. patent application 16/265,016 (the ‘016 patent application) on February 1, 2019 which claims priority to U.S. Provisional Patent Application Number 62/219,302, filed on September 16, 2015, and U.S. Provisional Patent Application Number 62/239,696, filed on October 9, 2015. This application was allowed on February 6, 2020, with claims specifically directed to a method for treating a coronavirus infection in a human with Remdesivir.
Patentability of Wuhan’s Patent Application for COVID-19 The patentability of Wuhan’s patent application for COVID-19 will turn on whether the application satisfies the requirements of patentability in the country that it is seeking the patent. These include i) novelty, ii) inventive step/obviousness, and iii) written description and enablement. The patent application has a good chance of satisfying the novelty requirement since COVID-19 is a novel coronavirus and Gilead’s patents do not specifically mention COVID-19. Likewise, written description will likely be satisfied assuming the patent application was filed with the necessary data and descriptions. The main hurdle to patentability will be inventiveness, or obviousness. Since Gilead’s patent specifically mentions using Remdesivir to treat viral infections caused by a Coronaviridae virus, the patent office could reject the claims because, arguably, it could have been obvious to one skilled in the art to use Remdesivir for any coronavirus, including COVID-19. To overcome this rejection, Wuhan will need to show that there is something unique or unexpected in its treatment methods, whether it is the dose, the dosing regimen, or even the safety profile. Overall, obtaining a patent will not be automatic for Wuhan but it is certainly possible that they will receive a patent with claims narrowly tailored to treating COVID-19. Freedom to Operate (FTO) Assuming Wuhan is able to receive a patent, the next question is whether it will be able to market or sell the treatment. This comes down to whether or not Wuhan has freedom to operate, or FTO. As the name implies, this analysis requires determining whether there are any blocking patents in the geographic region in which you plan to commercialize (i.e., make, sell, distribute, use, and otherwise market) the indicated therapy. That is, in order to determine whether or not Wuhan can commercialize the COVID-19 therapy, you have to see whether that therapy falls within the scope of any third-party patent rights within the jurisdiction of interest. In this case, the potential blocking IP would be Gilead’s patent for Remdesivir. Looking at the U.S. claims, Gilead’s ‘958 patent broadly claims the composition. It is not limited by any additional features. As such, the ‘958 patent gives Gilead rights to Remdesivir for all uses. Since Wuhan’s patent application relies on Remdesivir, it’s therapy would fall within the scope of the ‘958 patent. Moreover, Wuhan’s therapy would likely also run afoul of Gilead’s ‘016 patent application which is directed to the method of treating coronaviridae infection with Remdesivir. Since COVID-19 is a specific coronavirus, any methods of “treating COVID-19” would likely fall within the scope of “treating coronaviridae infection.” Just as with the ‘958 patent, Wuhan’s application of using Remdesivir to treat COVID-19 infections would encounter obstacles with the ‘016 patent application. Conclusion As you can see, the questions of how Wuhan can patent a treatment for COVID-19 using Gilead’s pre-existing technology involves understanding the differences between being able to patent an invention and being able to actually commercialize it. Of course, finding that certain patent rights exist that could block commercialization of Remdisivir for the treatment of COVID-19 does not necessarily end the analysis. Designing around Gilead’s patents, licensing Gilead’s patents, or even obtaining a compulsory license in this situation can help Wuhan overcome the patent hurdles to commercialization.
0 Comments
Leave a Reply. |
Welcome!BioPharma Law Blog posts updates and analyses on IP topics, FDA regulatory issues, emerging legal developments, and other news in the constantly evolving world of biotech, pharma, and medical devices. Archives
March 2021
Categories
All
|
Practices |
Company
|
|

 RSS Feed
RSS Feed